Benjamin Franklin to Ebenezer Kinnersley, 17 July 1771
To Ebenezer Kinnersley
ALS: University of Pennsylvania Library
London, July 17. 1771
Dear Sir,
I was much oblig’d by your Account of the Effect of the Lightning on Mr. Holder’s6 House. It will be in the Transactions here. I wonder it is not to be found in yours.
Those here who aimed at obtaining a very great Electric Force, have been much discouraged by the Breaking of the Bottles that compos’d their Batteries. A Gentleman of my Acquaintance lost 8 out of 20 at one Stroke; another 12 out of 40.7 Having heard that Pere Beccaria had lined the Inside of a great Iron Kettle with Cement, and then coated a Part of the Cement with Tinfoil, from whence he could discharge a great Stroke; and if any Crack happen’d to his Cement, he mended it again with a hot Iron; I recommended trying to make Batteries of Paper, by straining the Sheets on Frames, drying them hot before the Fire, then impregnating them with melted Wax, and afterwards coating them with Tinfoil.8 This another ingenious Friend has try’d, and as he writes me it succeeds.
The same (Mr. Henley) has invented an Electrometer which seems useful. I send you a Draft of it.9 It shows in what degree a Bottle is charg’d, that is, whether half, three quarters, &c. So that knowing the Force of a full Charge of any Bottle or Battery, you may by this, while charging, know the Proportion you have of such Force.
Your Experiment showing that a Stroke with black Lead on Paper would conduct a Shock, was new to me. I mention’d it to some who since tell me that they also find the solid Black Lead in a Pencil conducts as well as Wire; Which indeed, (the other being true) is not to be wonder’d at. It is however the only Property of Metal Black Lead possesses, as far as we yet know it.1
Mr. Canton melts Silver and Gold Wire by Electricity, not only into fine bright little Globules, but also into Spherules of Glass, some of which he has shown me by his Microscope. They were transparent, the Light passing thro’ them, and appearing in a Focus on the Paper.2
Mr. Henley has several Times melted Iron Wire lying at the Bottom of a White Stone Plate filled with Water. The Iron was destroy’d, and mark’d the Plate with an indelible black Stroke.
Sparks flew from it out thro’ the Water, and fell red hot on the Table.3
I wish I had any thing of more Importance to communicate. Business, during the Winter, takes up my time, so that I make no Experiments myself, but what I hear of I shall continue to send you. Being, with sincere Esteem, Dear Sir, Your most obedient humble Servant
B Franklin
P.S. I cannot find the Draft I promised, so must attempt a Sketch.
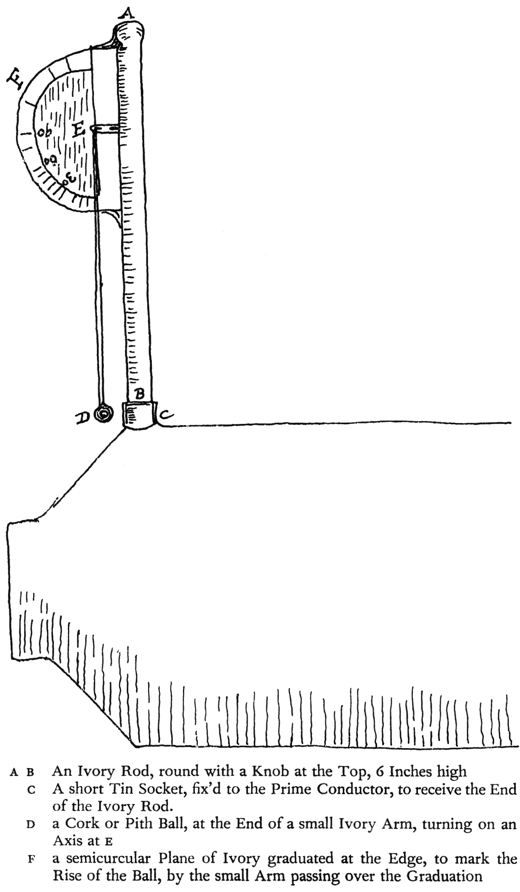
[The drawing is based on the original, which is too faint to reproduce.]
Mr Kinnersley
Endorsed: To Dr Ebenezer Kinnersley. From Dr. Benjamin Franklin
6. A misspelling of Moulder; see above, XVII, 249.
7. Experimenters, partly in the hope of simulating the effects of lightning, were trying to build the largest possible charges in their batteries, which were commonly made by connecting a number of Leyden jars; see above, IV, 202. Breaking of the jars was a widespread problem, to which Edward Nairne and Giambatista Beccaria eventually published solutions: Phil. Trans., LXIV (1774), 87; Beccaria, A Treatise upon Artificial Electricity... (London, 1776), pp. 118–21. Either of these men may have been one of the acquaintances to whom BF refers. The other was unquestionably Priestley; BF and Canton were with him in June, 1771, when several of his jars exploded. Priestley, History, II, 242–6, and Experiments and Observations Relating to Various Branches of Natural Philosophy (3 vols., London, 1779–86), II, 286–7.
8. BF presumably heard of Beccaria’s innovation from Priestley, who was also experimenting with cement (ibid., pp. 287–91); how the Italian could have mended cement with a hot iron we do not venture to guess. BF’s suggestion of paper for batteries was a development of the glass sheets, covered with thin lead, that he had used many years before: above, III, 357–8.
9. For another drawing and a description of Henly’s electrometer see above, XVII, 259–61.
1. Black lead or graphite is a form of carbon which, unlike the diamond, is an excellent conductor. Priestley was experimenting with another conductive form, charcoal: History, II, 193–200.
2. Gold and silver, like other metals, disintegrate when exposed to a sufficient charge, and form small particles that reflect brilliantly. Although the gold in fact remains gold and the silver silver, the metals were widely thought to be changed—as if by alchemy in reverse—into some kind of glass-like material. See ibid., p. 292.
3. Henly, we conjecture, was trying to improve the construction of lightning rods. Priestley was conducting experiments along similar lines, from which he concluded that an electrical discharge affected wet metals more than dry. Ibid., p. 271; see also pp. 251–2, 263–4.

![University of Virginia Press [link will open in a new window] University of Virginia Press](/lib/media/rotunda-white-on-blue.png)
